Marc Baumgardner, PhD
News
Post: We've just had an article accepted to Combustion and Flame, which is (arguably) the premiere journal for combustion science and is the official journal of the Combustion Institute. In some respects, this paper has been a long time coming in that the majority of the buildup of the experimental apparatus and much of the physical data-taking was done by students here at GU over the past few years. I'm so proud of all of the students that have passed through the lab and especially of John Harvey, the co-author on this particular paper. John began working in the lab this past year and his work ethic and dedication have been truly remarkable. Thanks John!
Marc E. Baumgardner and John Harvey
Title: Analyzing OH*, CH*, and C2* chemiluminescence of bifurcating FREI propane-air flames in a micro flow reactor
Abstract: An experimental investigation of spontaneous flame chemiluminescence of OH*, CH*, and C2* radicals was conducted for premixed propane-air flames in a micro flow reactor. Equivalence ratios from 0.7 to 1.4 were examined and correlated well to the ratio of OH*/CH* and C2*/CH* (R2 > 0.999 for C2*/CH* power fit). The OH*/CH* ratio better correlated with equivalence ratio for lean flames, while the C2*/CH* ratio better correlated with rich flames. The equivalence ratio model was used to calculate the axial local equivalence ratio for a rich propane-air flame (phi of 1.5) exhibiting strong bifurcating FREI behavior, resulting in three visible flame fronts. The measured intensities of OH*, CH*, and C2* chemiluminescence, as well as the calculated local equivalence ratio along the reactor axis support the numerical model explanations of the observed bifurcating FREI flames available in the literature. This work highlights the novel application of this diagnostic technique in the context of micro flow reactors.
Marc E. Baumgardner and John Harvey
Title: Analyzing OH*, CH*, and C2* chemiluminescence of bifurcating FREI propane-air flames in a micro flow reactor
Abstract: An experimental investigation of spontaneous flame chemiluminescence of OH*, CH*, and C2* radicals was conducted for premixed propane-air flames in a micro flow reactor. Equivalence ratios from 0.7 to 1.4 were examined and correlated well to the ratio of OH*/CH* and C2*/CH* (R2 > 0.999 for C2*/CH* power fit). The OH*/CH* ratio better correlated with equivalence ratio for lean flames, while the C2*/CH* ratio better correlated with rich flames. The equivalence ratio model was used to calculate the axial local equivalence ratio for a rich propane-air flame (phi of 1.5) exhibiting strong bifurcating FREI behavior, resulting in three visible flame fronts. The measured intensities of OH*, CH*, and C2* chemiluminescence, as well as the calculated local equivalence ratio along the reactor axis support the numerical model explanations of the observed bifurcating FREI flames available in the literature. This work highlights the novel application of this diagnostic technique in the context of micro flow reactors.
Here's a pre-print of the accepted article.
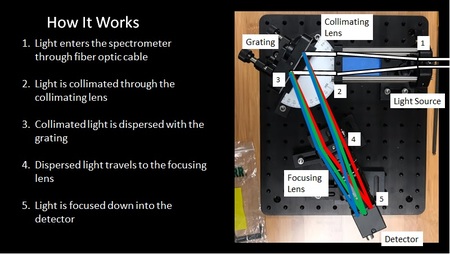
Post: This past year's senior design project for the lab was the design and construction of a Raman Spectrometer -- the goal being to measure species within the micro-reactor and linear Hencken-type burner. Here is a link to their senior design presentation with some details. The goal for this summer is to start collecting data and incorporate this new tool into our existing experiments.
Post: The new burner is finally done!! It is a linear Hencken-type burner and is modeled after the one built by the group at Sandia National Labs: https://aip.scitation.org/doi/10.1063/1.4967491


Post: Senior design day. The team presenting their Hybrid Rocket Engine Test Stand

Post: Engine skid up an running. This test stand was constructed by a senior design team, and should be good for engines up to 150 hp.

Post: It's been a while, but that's how things go during the school year. Research is continuing and I have a new student working in the lab, which is great! We recently presented our work at a research colloquium here on campus. Below is a (small) picture - click on it to download the actual poster (*.pdf).

Post: This latest article is pretty special; it was work led by Dr. Jessica Tryner at CSU and one of, if not the most thorough investigations on the impact that secondary air injection has on exhaust emissions from gassifier cookstoves. This project was funded by a U.S.DOE grant and sought to achieve Tier 4 emissions from a gassifier cookstove (which we met for certain cooking phases). More simply, we set out to understand and create the most efficient, lowest emitting biomass cookstove possible. Developments from this work could go on to make cooking safer for, literally, billions of people. I'm very grateful for being able to contribute to this work and look forward to further investigations in this area.
Jessica Tryner, James W. Tillotson, Marc E. Baumgardner, Jeffrey T. Mohr, Morgan W. DeFoort, and Anthony J. Marchese
Title: The Effects of Air Flow Rates, Secondary Air Inlet Geometry, Fuel Type, and Operating Mode on the Performance of Gasifier Cookstoves
Abstract: Development of biomass cookstoves that reduce emissions of CO and PM2.5 by more than 50% and 95%, respectively, compared to a three-stone fire has been promoted as part of efforts to reduce exposure to household air pollution (HAP) among people that cook with solid fuels. Gasifier cookstoves have attracted interest because some have been shown to emit less CO and PM2.5 than other designs. A laboratory test bed and new test procedure were used to investigate the influence of air flow rates, stove geometry, fuel type, and operating mode on gasifier cookstove performance. Power output, CO emissions, PM2.5 emissions, fuel con- sumption rates, producer gas composition, and fuel bed temperatures were measured. The test bed emitted <41 mg· MJ − 1 PM2.5 and <8 g·MJd −1 CO when operating normally with certain prepared fuels, but order of magnitude increases in emission factors were observed for other fuels and during refueling. Changes in operating mode and fuel type also affected the composition of the producer gas entering the secondary combustion zone. Overall, the results suggest that the effects of fuel type and operator behavior on emissions need to be considered, in addition to cookstove design, as part of efforts to reduce exposure to HAP.
Post: This past year's senior design project for the lab was the design and construction of a Raman Spectrometer -- the goal being to measure species within the micro-reactor and linear Hencken-type burner. Here is a link to their senior design presentation with some details. The goal for this summer is to start collecting data and incorporate this new tool into our existing experiments.

Post: The new burner is finally done!! It is a linear Hencken-type burner and is modeled after the one built by the group at Sandia National Labs: https://aip.scitation.org/doi/10.1063/1.4967491


Post: Senior design day. The team presenting their Hybrid Rocket Engine Test Stand

Post: Engine skid up an running. This test stand was constructed by a senior design team, and should be good for engines up to 150 hp.

Post: It's been a while, but that's how things go during the school year. Research is continuing and I have a new student working in the lab, which is great! We recently presented our work at a research colloquium here on campus. Below is a (small) picture - click on it to download the actual poster (*.pdf).

Post: This latest article is pretty special; it was work led by Dr. Jessica Tryner at CSU and one of, if not the most thorough investigations on the impact that secondary air injection has on exhaust emissions from gassifier cookstoves. This project was funded by a U.S.DOE grant and sought to achieve Tier 4 emissions from a gassifier cookstove (which we met for certain cooking phases). More simply, we set out to understand and create the most efficient, lowest emitting biomass cookstove possible. Developments from this work could go on to make cooking safer for, literally, billions of people. I'm very grateful for being able to contribute to this work and look forward to further investigations in this area.
Jessica Tryner, James W. Tillotson, Marc E. Baumgardner, Jeffrey T. Mohr, Morgan W. DeFoort, and Anthony J. Marchese
Title: The Effects of Air Flow Rates, Secondary Air Inlet Geometry, Fuel Type, and Operating Mode on the Performance of Gasifier Cookstoves
Abstract: Development of biomass cookstoves that reduce emissions of CO and PM2.5 by more than 50% and 95%, respectively, compared to a three-stone fire has been promoted as part of efforts to reduce exposure to household air pollution (HAP) among people that cook with solid fuels. Gasifier cookstoves have attracted interest because some have been shown to emit less CO and PM2.5 than other designs. A laboratory test bed and new test procedure were used to investigate the influence of air flow rates, stove geometry, fuel type, and operating mode on gasifier cookstove performance. Power output, CO emissions, PM2.5 emissions, fuel con- sumption rates, producer gas composition, and fuel bed temperatures were measured. The test bed emitted <41 mg· MJ − 1 PM2.5 and <8 g·MJd −1 CO when operating normally with certain prepared fuels, but order of magnitude increases in emission factors were observed for other fuels and during refueling. Changes in operating mode and fuel type also affected the composition of the producer gas entering the secondary combustion zone. Overall, the results suggest that the effects of fuel type and operator behavior on emissions need to be considered, in addition to cookstove design, as part of efforts to reduce exposure to HAP.
DOI: 10.1021/acs.est.6b00440
Post: Success! What you see here is the first data from our micro-reactor experiment...very exciting! In the reactor is a stoichiometric methane/air flame flowing at about 50 cm/s; through the burner is a lean hydrogen-air flame.

And here the guys are making it happen!

Post: Hydrogen flame in action, note the thermal imaging camera picking up the flame (H2 burns invisible):

The guys have done a great job and we're getting really close to finally collecting some data!
Post: Methane-Air flame on the flat-flame burner...

Post: A minor delay in the gas bottles means that it'll be end of the week before we can actually run...however, the overall setup for both experiments is essentially complete (at least for initial testing purposes).

...and some graphical explanation of what's what:

more to come as preliminary data begins to roll in.
Post: Gauges!! We're getting quite close to running experiments. The guys have been working hard and it's all finally starting to come together. Gas bottles should arrive tomorrow - initial shakedown testing should begin next week!

Post: Summer research is moving along, we've got water and air hooked up and should be getting gas bottles in the next week or so. Once those are in we'll be ready to start with preliminary experiments...or as we're calling it in the lab "shooting lasers at fire" - more on that to come!
I thought I'd take a moment to talk about some effective use of 3-D printing in the laboratory environment. 3-D printing has come a long way in recent years; as an extreme case check out this story on Boeing's use of the technology as well as where they're potentially headed with it. Now, the Boeing case is an extreme one and likely well beyond the capabilities of smaller shops and laboratories. Additionally, the types of things and materials that can be printed by more affordable printers (say something in the $1000 range) will not have the precision or strength that may be required. For example, you are not likely going to give up purchasing from Swagelok in lieu of printing your own fittings anytime soon. However, I do believe that 3-D printing does have a place in the lab setting, especially when you're trying to keep experimental setup affordable. Honestly, when trying set up a low-cost experiment it's the little things that will get you; it's fairly easy to price out the big stuff and the main components, but trying to foresee every little item can be difficult...also it's sometimes difficult to accurately forecast exactly how you're going to anchor that manifold, or move that one piece in relation to that other piece (whatever they may be). It's in these instances where 3-D printing can really shine (and maybe even save the day)!
Here are some nice examples of how one of my students (WS) has been using our printers lately:
Door hardware: now we're probably only saving a few bucks on each one of these hinges and handles, but that adds up quickly. These cost maybe $6 to print, but would have cost us upwards of $60 for all of these plus tax and shipping. Plus, I really love the custom look and feel to them!


Other places where (affordable) 3-D printing can make sense is when you need custom machined parts, but the precision doesn't have to be quite there.
Manifold support: There are a few manifolds in our experiments and WS has printed up a really nice support structure for one of them:


One challenge we ran into with our MicroRx experiment was that we needed a way to slide a long thermocopule back and forth easily. We printed up this apparatus, combined with a few bucks in McMaster 80/20 and some 1/4" tubing, and voila(!) looks great (and works even better)!

So there you have it, some easy ways to put 3-D printing to good use. I've got some other ideas to use it for more research-oriented purposes, but more on that some other day.
Post: It's been a busy couple weeks; I traveled to CSU for some meetings with collaborators and then was on vacation for the 4th, but now I'm back in the lab and things are progressing...check out our new fume hood:

Post: Another article accepted! I'm really proud of this one as it is in the quite prestigious (at least in combustion circles) Proceedings of the Combustion Institute. This work was a fantastic collaboration with some folks back at CSU and involves laser ignition. What's really special about this work is that it's the first look at laser ignition in a RCM where conditions are both at high temperature and pressure (which is atypical of previous research in this area).
Ciprian Dumitrache, Marc Baumgardner, Andrew Boissiere, Amir Maria, John Roucis, Anthony J. Marchese, Azer Yalin
Title: A study of laser induced ignition of methane–air mixtures inside a Rapid Compression Machine
Abstract: Presented herein is a fundamental study of laser ignition of methane/air mixtures at temperatures and pressures representative of an internal combustion engine. An Nd:YAG laser operating at λ = 1064 nm was used to ignite methane/air mixtures at equivalence ratios of 0.4 ≤ Φ ≤ 1 in a Rapid Compression Machine (RCM). Experiments were conducted to study the lean limit, minimum spark energy (MSE), and minimum ignition energy (MIE). The results show that laser ignition exhibits a stochastic behavior which must be interpreted statistically. A 90% probability of occurrence was used to evaluate the MSE and MIE, which resulted in MSE90 =2.3 mJ and MIE90 =7.2 mJ at an equivalence ratio Φ = 0.4 at compressed pressure and temperature of Pcomp = 29 bar and Tcomp = 750 K, respectively. The lean limit was characterized based on the fraction of chemical energy converted into thermal energy, which was determined by calculating the apparent rate of heat release as derived from RCM high speed pressure data. A lean limit for 90% chemical energy conversion was found to correspond to an equivalence ratio of 0.47 (Tcomp = 782 K). Schlieren photography was employed as a diagnostics tool to visualize the flame initiation and propagation inside the RCM.
DOI: 10.1016/j.proci.2016.05.033
Post: Had to put a new head gasket on the B&S, looks like the spark plug may have been tightened just a bit too much...but no worries, this is a working lab and the little engine was back up and running in no time. We're now ready to start making pulls on the dyno!

Also, got an important delivery today: Thorlabs party-time!

Post: JC in the shop grinding away on some parts for this summer's project: getting the micro-reactor and LED-absorption experiments up and going.

Post: I currently have two great students working for me this summer setting up experiments and generally getting the laboratory in fighting shape, as it were.
Post: Presented a paper at the Western States Section of the Combustion Institute
https://www.engr.washington.edu/news/wssci2016
Post: Just had an article accepted!
M.E.Baumgardner et al. Energy & Fuels 29 (2015) 7317-7326
Title: Combustion of Lignocellulosic Biomass Based Oxygenated Components in a Compression Ignition Engine
Abstract: Processes such as fast pyrolysis of whole biomass or base-catalyzed depolymerization of lignin produce complex mixtures of oxygenated compounds that must be upgraded to be suitable for blending with petroleum and processing in a refinery. Complete removal of these oxygenated compounds is exceedingly energy intensive, and it is likely that upgraded pyrolysis oils will contain up to 2% oxygen content to be economically viable. The purpose of this study was to evaluate the effect of the presence of oxygenated chemical components representative of those present in upgraded pyrolysis oil on diesel engine performance and emissions. Engine testing was performed by blending seven different oxygenated components and one multicomponent blend with certification ultralow sulfur diesel fuel and quantifying the performance and emissions from the combustion of these fuels in a four-cylinder, turbocharged, 4.5 L John Deere PowerTech Plus common rail, direct injection diesel engine that meets Tier 3 off-highway emissions specifications. The properties of the oxygenated fuel components were fully characterized in accordance with ASTM diesel fuel standards. Gaseous emissions measurements included CO, CO2, NO, NO2, and total hydrocarbons; particulate measurements were performed on a PM10 basis. The residual oxygenate blends exhibited very few statistically significant differences compared to diesel at lower blend levels (2 vol %) but negative effects were observed at higher blend levels (5−6 vol %).
DOI: 10.1021/acs.energyfuels.5b01595
Post: Success! What you see here is the first data from our micro-reactor experiment...very exciting! In the reactor is a stoichiometric methane/air flame flowing at about 50 cm/s; through the burner is a lean hydrogen-air flame.

And here the guys are making it happen!

Post: Hydrogen flame in action, note the thermal imaging camera picking up the flame (H2 burns invisible):

The guys have done a great job and we're getting really close to finally collecting some data!
Post: Methane-Air flame on the flat-flame burner...

Post: A minor delay in the gas bottles means that it'll be end of the week before we can actually run...however, the overall setup for both experiments is essentially complete (at least for initial testing purposes).

...and some graphical explanation of what's what:

more to come as preliminary data begins to roll in.
Post: Gauges!! We're getting quite close to running experiments. The guys have been working hard and it's all finally starting to come together. Gas bottles should arrive tomorrow - initial shakedown testing should begin next week!

Post: Summer research is moving along, we've got water and air hooked up and should be getting gas bottles in the next week or so. Once those are in we'll be ready to start with preliminary experiments...or as we're calling it in the lab "shooting lasers at fire" - more on that to come!
I thought I'd take a moment to talk about some effective use of 3-D printing in the laboratory environment. 3-D printing has come a long way in recent years; as an extreme case check out this story on Boeing's use of the technology as well as where they're potentially headed with it. Now, the Boeing case is an extreme one and likely well beyond the capabilities of smaller shops and laboratories. Additionally, the types of things and materials that can be printed by more affordable printers (say something in the $1000 range) will not have the precision or strength that may be required. For example, you are not likely going to give up purchasing from Swagelok in lieu of printing your own fittings anytime soon. However, I do believe that 3-D printing does have a place in the lab setting, especially when you're trying to keep experimental setup affordable. Honestly, when trying set up a low-cost experiment it's the little things that will get you; it's fairly easy to price out the big stuff and the main components, but trying to foresee every little item can be difficult...also it's sometimes difficult to accurately forecast exactly how you're going to anchor that manifold, or move that one piece in relation to that other piece (whatever they may be). It's in these instances where 3-D printing can really shine (and maybe even save the day)!
Here are some nice examples of how one of my students (WS) has been using our printers lately:
Door hardware: now we're probably only saving a few bucks on each one of these hinges and handles, but that adds up quickly. These cost maybe $6 to print, but would have cost us upwards of $60 for all of these plus tax and shipping. Plus, I really love the custom look and feel to them!


Other places where (affordable) 3-D printing can make sense is when you need custom machined parts, but the precision doesn't have to be quite there.
Manifold support: There are a few manifolds in our experiments and WS has printed up a really nice support structure for one of them:


One challenge we ran into with our MicroRx experiment was that we needed a way to slide a long thermocopule back and forth easily. We printed up this apparatus, combined with a few bucks in McMaster 80/20 and some 1/4" tubing, and voila(!) looks great (and works even better)!

So there you have it, some easy ways to put 3-D printing to good use. I've got some other ideas to use it for more research-oriented purposes, but more on that some other day.
Post: It's been a busy couple weeks; I traveled to CSU for some meetings with collaborators and then was on vacation for the 4th, but now I'm back in the lab and things are progressing...check out our new fume hood:

Post: Another article accepted! I'm really proud of this one as it is in the quite prestigious (at least in combustion circles) Proceedings of the Combustion Institute. This work was a fantastic collaboration with some folks back at CSU and involves laser ignition. What's really special about this work is that it's the first look at laser ignition in a RCM where conditions are both at high temperature and pressure (which is atypical of previous research in this area).
Ciprian Dumitrache, Marc Baumgardner, Andrew Boissiere, Amir Maria, John Roucis, Anthony J. Marchese, Azer Yalin
Title: A study of laser induced ignition of methane–air mixtures inside a Rapid Compression Machine
Abstract: Presented herein is a fundamental study of laser ignition of methane/air mixtures at temperatures and pressures representative of an internal combustion engine. An Nd:YAG laser operating at λ = 1064 nm was used to ignite methane/air mixtures at equivalence ratios of 0.4 ≤ Φ ≤ 1 in a Rapid Compression Machine (RCM). Experiments were conducted to study the lean limit, minimum spark energy (MSE), and minimum ignition energy (MIE). The results show that laser ignition exhibits a stochastic behavior which must be interpreted statistically. A 90% probability of occurrence was used to evaluate the MSE and MIE, which resulted in MSE90 =2.3 mJ and MIE90 =7.2 mJ at an equivalence ratio Φ = 0.4 at compressed pressure and temperature of Pcomp = 29 bar and Tcomp = 750 K, respectively. The lean limit was characterized based on the fraction of chemical energy converted into thermal energy, which was determined by calculating the apparent rate of heat release as derived from RCM high speed pressure data. A lean limit for 90% chemical energy conversion was found to correspond to an equivalence ratio of 0.47 (Tcomp = 782 K). Schlieren photography was employed as a diagnostics tool to visualize the flame initiation and propagation inside the RCM.
DOI: 10.1016/j.proci.2016.05.033
Post: Had to put a new head gasket on the B&S, looks like the spark plug may have been tightened just a bit too much...but no worries, this is a working lab and the little engine was back up and running in no time. We're now ready to start making pulls on the dyno!

Also, got an important delivery today: Thorlabs party-time!

Post: JC in the shop grinding away on some parts for this summer's project: getting the micro-reactor and LED-absorption experiments up and going.

Post: I currently have two great students working for me this summer setting up experiments and generally getting the laboratory in fighting shape, as it were.
Post: Presented a paper at the Western States Section of the Combustion Institute
https://www.engr.washington.edu/news/wssci2016
Post: Just had an article accepted!
M.E.Baumgardner et al. Energy & Fuels 29 (2015) 7317-7326
Title: Combustion of Lignocellulosic Biomass Based Oxygenated Components in a Compression Ignition Engine
Abstract: Processes such as fast pyrolysis of whole biomass or base-catalyzed depolymerization of lignin produce complex mixtures of oxygenated compounds that must be upgraded to be suitable for blending with petroleum and processing in a refinery. Complete removal of these oxygenated compounds is exceedingly energy intensive, and it is likely that upgraded pyrolysis oils will contain up to 2% oxygen content to be economically viable. The purpose of this study was to evaluate the effect of the presence of oxygenated chemical components representative of those present in upgraded pyrolysis oil on diesel engine performance and emissions. Engine testing was performed by blending seven different oxygenated components and one multicomponent blend with certification ultralow sulfur diesel fuel and quantifying the performance and emissions from the combustion of these fuels in a four-cylinder, turbocharged, 4.5 L John Deere PowerTech Plus common rail, direct injection diesel engine that meets Tier 3 off-highway emissions specifications. The properties of the oxygenated fuel components were fully characterized in accordance with ASTM diesel fuel standards. Gaseous emissions measurements included CO, CO2, NO, NO2, and total hydrocarbons; particulate measurements were performed on a PM10 basis. The residual oxygenate blends exhibited very few statistically significant differences compared to diesel at lower blend levels (2 vol %) but negative effects were observed at higher blend levels (5−6 vol %).
DOI: 10.1021/acs.energyfuels.5b01595
